It has also been reported that daily temperature could affect the risk of disease transmission. In Egypt, it has been reported that H5N1 outbreaks in bird populations occur in close temporal proximity to human H5N1 cases, with human cases generally occurring within 30 days of an initial bird outbreak. These events may increase the likelihood global pandemic among humans. Furthermore, continued transmission of the pathogen between birds (and from birds) to humans has the potential to drive virus evolution with the possibility of increased virulence, and human-to-human transmission. 
Along with being a menace to human health, the continued presence of the pathogen in Egypt impacts the country’s protein sources, farmer’s livelihoods, the economy as a whole. In addition to bird cases, 168 humans in Egypt have been confirmed as infected with H5N1 as of August, 2012, and 60 of those cases resulted in death due to the disease. These outbreaks have caused the death of millions of birds due to illness or due to culling to prevent further spread of the disease. Outbreaks of H5N1 among small farming operations have remained prevalent over time, despite control measures. H5N1 was first detected in poultry populations in Egypt in February of 2006 and was shortly afterward found in 15 governorates around the country. Therefore, an important application of new, improved predictive models should be related to the emergence of zoonotic disease outbreaks in animals that could constitute health risk to nearby human populations. An example is the recent emergence of highly pathogenic avian influenza. More than two thirds of emerging infectious diseases in recent decades are zoonotic in origin (crossing from animals to humans). 
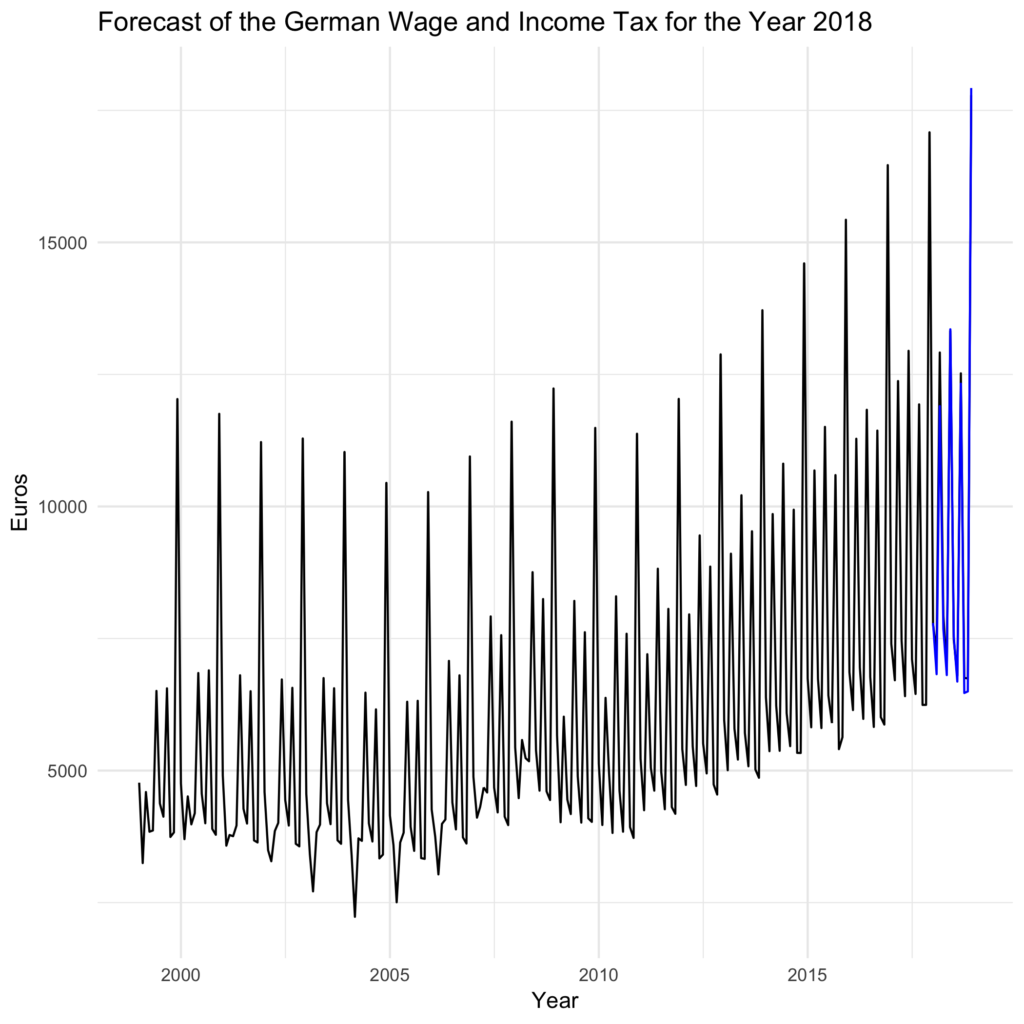
For such prevention to take place, timely and accurate prediction of outbreaks is critical. Examples of such preventive steps include vector control, public health messaging to avoid high-risk behaviors or areas, and raising clinician awareness for early diagnosis and treatment. By understanding when a future outbreak may occur, preventative steps can be taken to minimize its impact. Our analysis uncovers the time-series structure of outbreak severity for highly pathogenic avain influenza (H5N1) in Egypt.Ĭertain infectious diseases and disease events occur in a cyclic or rhythmic pattern related to climate or other factors that allows for modeling and prediction of future outbreaks. 2012), provides a new approach to predicting these dangerous outbreaks in bird populations based on existing, freely available data. This result, along with those showing the concordance between bird and human outbreaks (Rabinowitz et al. Random Forest time series modeling provides enhanced predictive ability over existing time series models for the prediction of infectious disease outbreaks. Furthermore, we found that the Random Forest model is effective for predicting outbreaks of H5N1 in Egypt. We found that the Random Forest model outperformed the ARIMA model in predictive ability. We applied ARIMA and Random Forest time series models to incidence data of outbreaks of highly pathogenic avian influenza (H5N1) in Egypt, available through the online EMPRES-I system. Developments in modeling approaches provide an opportunity to compare different time series models for predictive power. Incidence data can be used to predict the future occurrence of disease events. Time series models can play an important role in disease prediction. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
